Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Will Expand HCQ Protocol, Use it For Therapy & Prophylaxis: Niti Aayog's VK Paul | COVID-19 Updates - YouTube
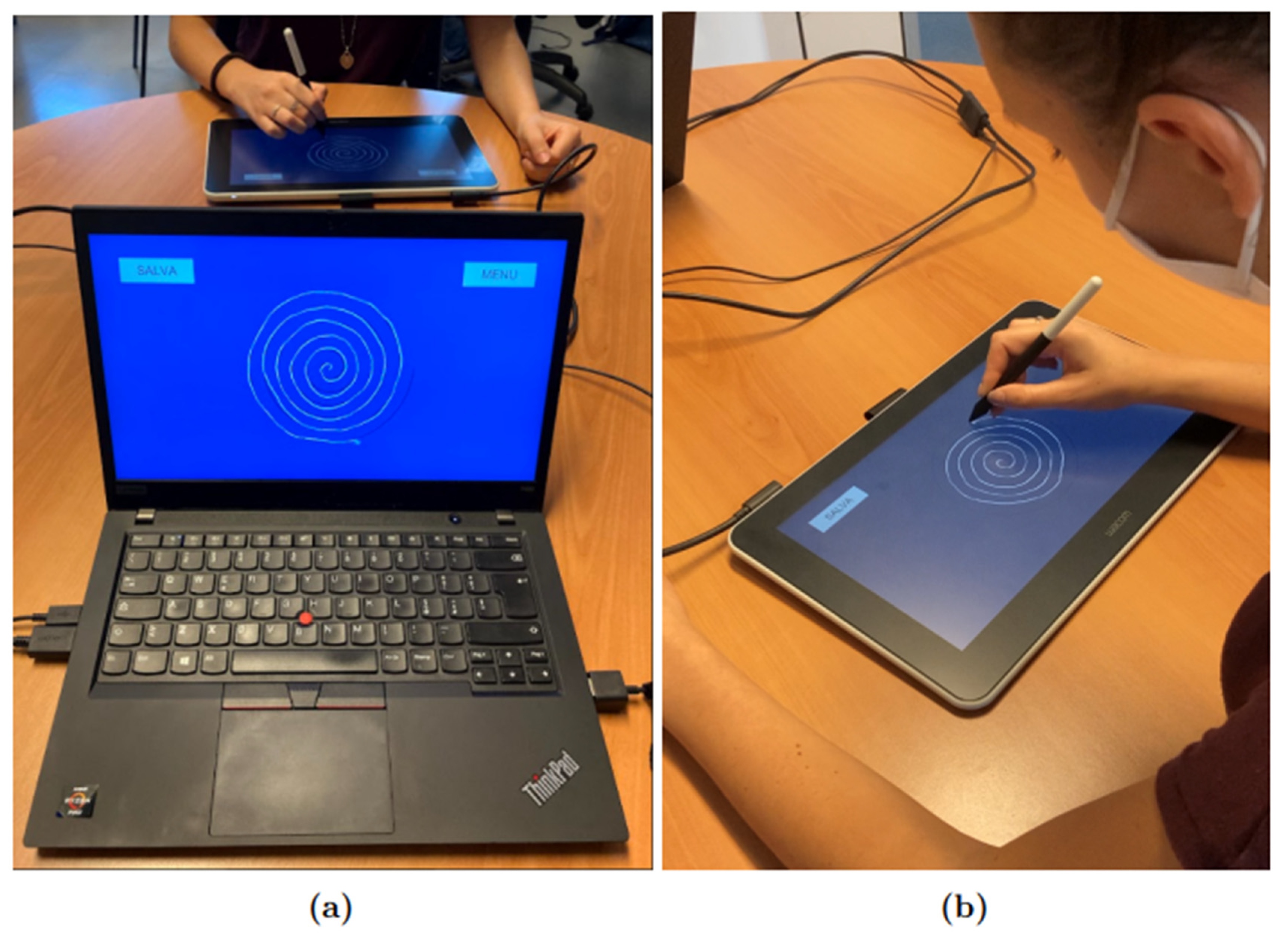
Engineering Proceedings | Free Full-Text | Classification-Based Screening of Parkinson’s Disease Patients through Graph and Handwriting Signals

Initial treatment with a single pill containing quadruple combination of quarter doses of blood pressure medicines versus standard dose monotherapy in patients with hypertension (QUARTET): a phase 3, randomised, double-blind, active-controlled trial -

Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial - The Lancet

Appendix C - Rapid Assessment of Roadside Habitat for Monarchs: Field Protocol and Datasheet | Evaluating the Suitability of Roadway Corridors for Use by Monarch Butterflies |The National Academies Press
Example of a Medication Dispensing Protocol to Accompany the Medication Assessment Form Developed by the Missouri Medical Counte

PDF) Extended-release pharmacotherapy for opioid use disorder (EXPO): protocol for an open-label randomised controlled trial of the effectiveness and cost-effectiveness of injectable buprenorphine versus sublingual tablet buprenorphine and oral liquid ...